Periodic Trends
Vocabulary
- atomic radius: half the radius between the toe nuclei of a diatomic molecule
- diatomic molecules: the 7 elements that always come in pairs (see above)
- ion: an atom with a positive or negative charge after it has gained or lost an electron
- trend: a specific pattern in the elements of the periodic table
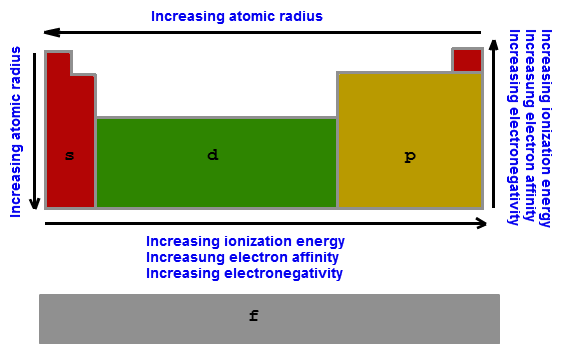
Periodic Table Trends
-vertical: larger from top to bottom
-vertical: decreases as you move down
-vertical: decreases as you go down
-vertical: increases as you move down
- atomic radius: the size of an atom from the nucleus to the electrons
-vertical: larger from top to bottom
- ionization energy: the energy it takes to remove an electron
-vertical: decreases as you move down
- electronegativity: the ability to attract an electron and form a covalent bond
-vertical: decreases as you go down
- ionic radius: the radius of an atoms ion
-vertical: increases as you move down